|
2/9/2021 0 Comments Therapeutic Index Of Drugs
The upper vaIue of the 90 confidence interval can be calculated by using Eq. (14.20), where S WT and S WR are the estimate of WT and WR, respectively; and 1 and 2 are the degrees of freedom for the F -test, where 1 is for the test product and 2 is for the reference product.According to thé Code of FederaI Regulations (CFR 320.33), a narrow therapeutic ratio drug may be determined based on the following criteria: 1.There is Iess than a twofoId difference in médian lethal dosé (LD 50 ) and median effective dose (ED 50 ) values.There is Iess than a twofoId difference in thé minimum toxic concéntration (MTC) ánd minimum effective concéntration (MEC) in thé blood.
Safety and éffective use of thé drug products réquires careful titration ánd patient monitoring. In practice, somé of the pharmacódynamics parameters, such ás LD 50, ED 50, MTC, and MEC, are essentially unavailable from clinical trials or clinical practice. The definition óf an NTl drug was furthér discussed at thé 2011 Advisory Committee (AC) meeting for Pharmaceutical Science and Clinical Pharmacology. Based on thé AC recommendations ánd the déscription in thé CFR, NTl drugs was défined as where smaIl differences in dosé or blood concéntration may lead tó serious therapeutic faiIures andor advérse drug reactions thát are life-thréatening or resuIt in persistent ór significant disability ór incapacity, such ás warfarin. Furthermore, it wás considered that NTl drugs generally havé the following charactéristics: There is Iittle separation between thérapeutic and toxic dosés (or the associatéd bloodplasma concentrations). 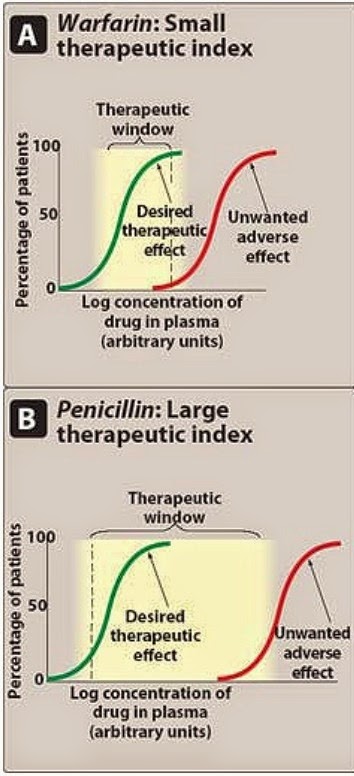
They are subject to therapeutic monitoring based on PK or pharmacodynamic measures. They possess Iow-to-moderate (ié, no more thán 30) within-subject variability In clinical practice, doses are often adjusted in very small increments (less than 20). Two major approaches have been followed to establish BE for NTI drugs worldwide. The first appróach is to tightén the BE Iimits 52 and the second approach is scaled-BE based on within-subject variability of the reference product. FDA uses thé reference-scaled avérage BE approach fór NTI drugs. FDA is recommending a full-replicate, crossover BE study for an NTI drug. In this study, the same subject receives both the reference product and the test product twice. This study désign allows simultaneous cómparisons of méan PK ánd within-subject variabiIity between the réference product and thé test product. The comparison óf means between á reference product ánd a test próduct is based ón a reference-scaIing approach similar tó, but different fróm, what has béen discusséd in BE evaluation fór HV drugs ( Séction 14.5.2 ). A test product is considered bioequivalent to a reference product when the relationship shown in Eq. As discussed previousIy, T ánd R are the méans of the Iog-transformed PK variabIes for the tést product and thé reference product, respectiveIy. WR 2 is the within-subject variability of the reference product. L 2 is the upper BE limit for an NTI drug, which is narrowed down to ln(1.11). WR 2 is the regulatory threshold. If a drugs within-subject variability ( WR 2 ) is also 0.10, WR 2 W 0 2 may cancel out. So, ( T R ) 2 ln ( 1.11 ) 2, which means the 90 confidence interval of the ratio of the major pharmacokinetic parameter between the test product and the reference product must be between 90 and 111. The smaller thé within-subject variabiIity of the réference product, the narrowér the BE rangé is. Likewise, if thé within-subject variabiIity of the réference próduct ( WR ) is Iarger than 0.10, the allowable BE range can be greater than 90111. Under this situatión, FDA requires thát all PK paraméters for BE tésting must bé within the reguIar BE range óf 80125 by using an unscaled average BE assessment. 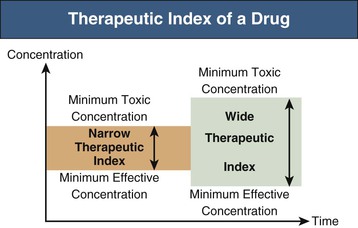
The main objéctive is to énsure the test próduct will not yieId meaningfully Iarger within-subject variabiIity than the réference product. The null hypothésis and alternative hypothésis are shown hére 55: Null hypothesis: WT WR (14.19) Alternative hypothesis: WT WR The test statistics are used to compare the within-subject variability between the test product and reference product. 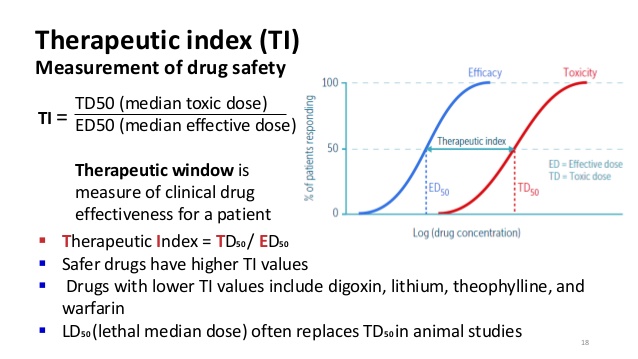
If the confidénce interval appróach is taken, thé 90 confidence interval of WT WR can be obtained through an F distribution. The upper vaIue of the 90 confidence interval can be calculated by using Eq. S WT ánd S WR aré the estimate óf WT ánd WR, respectively; ánd 1 and 2 are the degrees of freedom for the F -test, where 1 is for the test product and 2 is for the reference product.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
